Horii T, Morita S, Kimura M, Terawaki N, Shibutani M, Hatada I (Lab. of Genome Science, IMCR, Gunma Univ.)
About
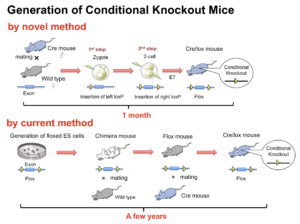
Conditional knockout using Cre/lox is essential for functional analysis of genes. CRISPR/Cas in combination with two sets of guide RNAs and a single-stranded oligonucleotide enables simultaneous insertion of two lox sequences. However, this method induces double-strand breaks at two sites on the same chromosome, which causes an undesirable chromosomal deletion and reduces the flanked lox (flox) rate. To solve this problem, we investigated a method that sequentially introduces each lox sequence at the 1-cell and 2-cell embryonic stages, respectively. The sequential method was applied to both microinjection and electroporation systems. Sequential electroporation improved the flox efficiency compared with ordinary simultaneous microinjection, leading to a high yield of offspring with floxed alleles. Finally, we directly produced Cre/lox mice containing both the Cre transgene and floxed allele via sequential electroporation using Cre zygotes, which accelerated the generation of conditional knockout mice compared with the ordinary method.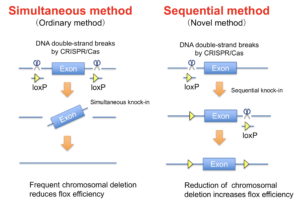
Paper information
Efficient generation of conditional knockout mice via sequential introduction of lox sites. Horii T, Morita S, Kimura M, Terawaki N, Shibutani M, Hatada I.
Sci Rep. 2017 Aug 11;7(1):7891. doi: 10.1038/s41598-017-08496-8. PMID: 28801621
Online URL
https://www.ncbi.nlm.nih.gov/pubmed/28801621